Product Database Manual
Contents
Product Databases
Pharmacy Simulator includes several product databases, appropriate to various regions around the world. This allows our international group of users to write and tailor their scenarios within the context of their local working conditions. It also allows our users to get a taste of what it's like to practice pharmacy in regions other than their own.
However, as much as we might try, it is unfortunately not possible or practical for us to maintain an up to date and comprehensive database of all medicinal products across all regions.
To work around this limitation, we provide tools which allow users to suggest additions and alterations to the various product databases that are included in Pharmacy Simulator. This way, if there is an important product that is missing you have the power to add the product yourself. Once approved, this product will become available for all users.
To begin making changes to the product database, open the Main Menu by clicking the Main Menu button, or pressing the Escape key, then click "Edit Product Database".
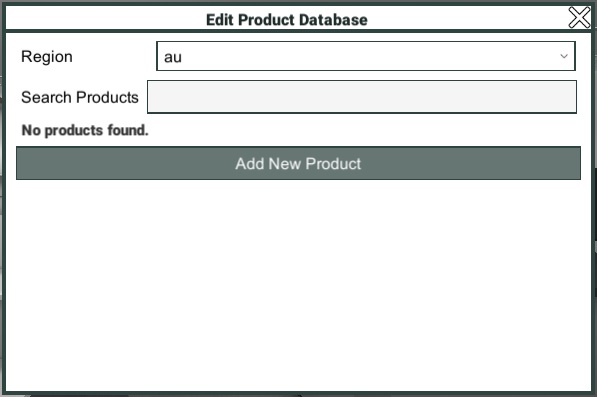
Edit Product Database
Having opened the Edit Product Database window, you will be able to either click the "Add New Product" button, to add a new product to the database. Or search for an existing product to make changes to. You can also change region, if you find yourself with the wrong product database loaded.
Note: You can only have one product database loaded at a time. If you switch regions it will take a moment to load the new product database. If you also have a scenario loaded which is incompatible with the selected region, the scenario will be closed.
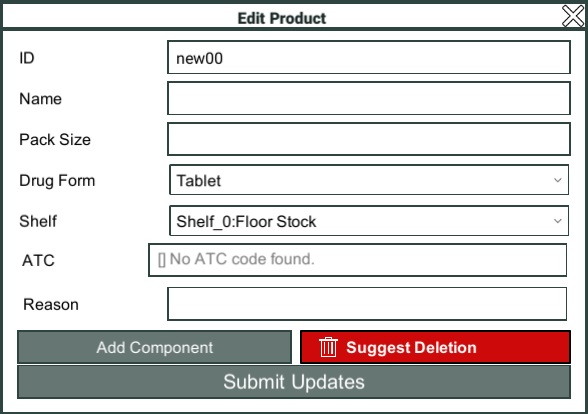
Add/Edit Product
Having clicked the Add Product button, or chosen an existing product to edit, the Edit Product window will open.
The ID field allows you to set a region unique identifier for the product. This can only be set if you are adding a new product.
The Name field allows you to enter the brand name of the product.
The Pack Size field allows you to enter how large the packet should be, if the product is sold in packets.
The Drug Form dropdown allows you to define what type of product it is, i.e. tablet, capsule, syrup, etc.
The Shelf dropdown allows you to define where in the Pharmacy the product would be stored.
The ATC field allows you to search for the correct ATC code for the product.
Note: The ATC code you select should be the most specific ATC code that is appropriate for the product you are adding. i.e. if you are adding a combination product, you should select the appropriate combination code, if one exists.
The Reason field allows you to enter a note to the reviewer to state why you think the addition/edit is required.
Once you have entered the basic details of the product, you need to define the generic components that make up the product. Most products will only have one component i.e. paracetamol, but some products may have multiple components i.e. paracetamol and codeine.
Click the "Add Component" button to add a component.
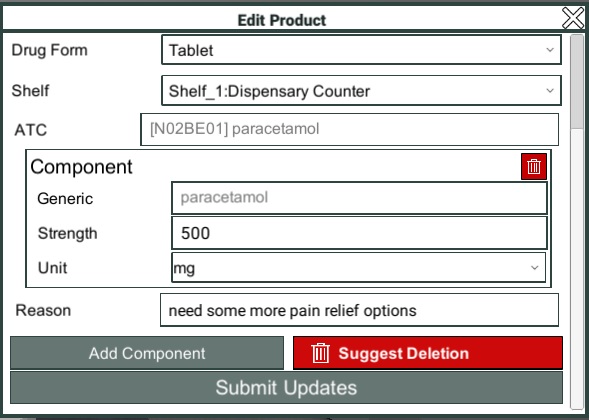
Add/Edit Component
Having clicked the Add Component button, or chosen a component to edit, you will see a form which allows you to select the component.
You can search for a generic ingredient and select from the comprehensive list of available options.
The Strength field allows you to define how much of the generic ingredient is present in the drug.
The Units dropdown allows you to specify what unit of measurement is used to measure the ingredient, i.e. mg, mL, etc.
Once you have finished making your edits, you can click the "Submit Updates" button. This will send your suggested changes through to moderators for review. They will determine if the product you are adding is appropriate, and either accept or reject your suggestion on this basis.
Once you have finished making your edits, you can click the "Submit Updates" button. This will send your suggested changes through to moderators for review. They will determine if the product you are adding is appropriate, and either accept or reject your suggestion on this basis.
In some cases the moderator may make some minor corrections, i.e. to fix a mistake in the product entry.
If your suggested change is critically wrong, i.e. it misses key components, or has incorrectly listed values, it may be rejected outright. Please try to ensure the products you suggest are appropriate, real, and useful.